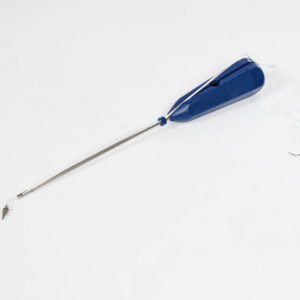
Bioabsorbable Interference Screws
Biocomposite/bioactive (PLGA + TCP) interference screw designed to provide the best involvement of soft tissue grafts (ACL-PCL) in tibia/femur without damaging the soft tissue in bone during cruciate ligament reconstruction procedures;
Thanks to its wide diameter and height options, INTAS provides possibilities of use additionally for, other than ACL – PCL reconstructions, repair of medial and lateral ligaments in the knee, medial patellafemoral ligament reconstruction (femur fixation), biceps tendonitis on the shoulder, and distal biceps tendonitis in the elbow, and makes it easier for the soft tissue to penetrate better into the bone by turning into bone tissue.
Thanks to ratcheting transmitter, it provides convenience for the doctor.
ORDERING INFORMATION:
| # | REF CODE | Description | SIZES |
|---|---|---|---|
| 1 | BAP19020625 | Intas | Ø6 mm x L25 mm |
| 2 | BAP19020630 | Intas | Ø6 mm x L30 mm |
| 3 | BAP19020725 | Intas | Ø7 mm x L25 mm |
| 4 | BAP19020730 | Intas | Ø7 mm x L30 mm |
| 5 | BAP19020825 | Intas | Ø8 mm x L25 mm |
| 6 | BAP19020830 | Intas | Ø8 mm x L30 mm |
| 7 | BAP19020925 | Intas | Ø9 mm x L25 mm |
| 8 | BAP19020930 | Intas | Ø9 mm x L30 mm |
| 9 | BAP19021025 | Intas | Ø10 mm x L25 mm |
| 10 | BAP19021030 | Intas | Ø10 mm x L30 mm |
| 11 | BAP19021125 | Intas | Ø11 mm x L25 mm |
| 11 | BAP19021130 | Intas | Ø11 mm x L30 mm |